MoCRA deadlines you need to keep in mind when selling cosmetic products in the U.S


MoCRA deadlines you need to keep in mind when selling cosmetic products in the U.S

Obelis Wins a Second Trends Gazelle Award.Growing from Medium-Sized to Big Enterprises CategoryWant to know more? Visit our page!

Manufacturers! Interested in the European Commission proposal for legacy devices? Our consultant will be offering an interpretation of the European Commission proposal for legacy devices regarding the following date: March 20, 2023

A definitive guide to CE marking in Europe. Discover what a CE marking is, what products require a CE mark, and why a CE mark is required on the EU market.
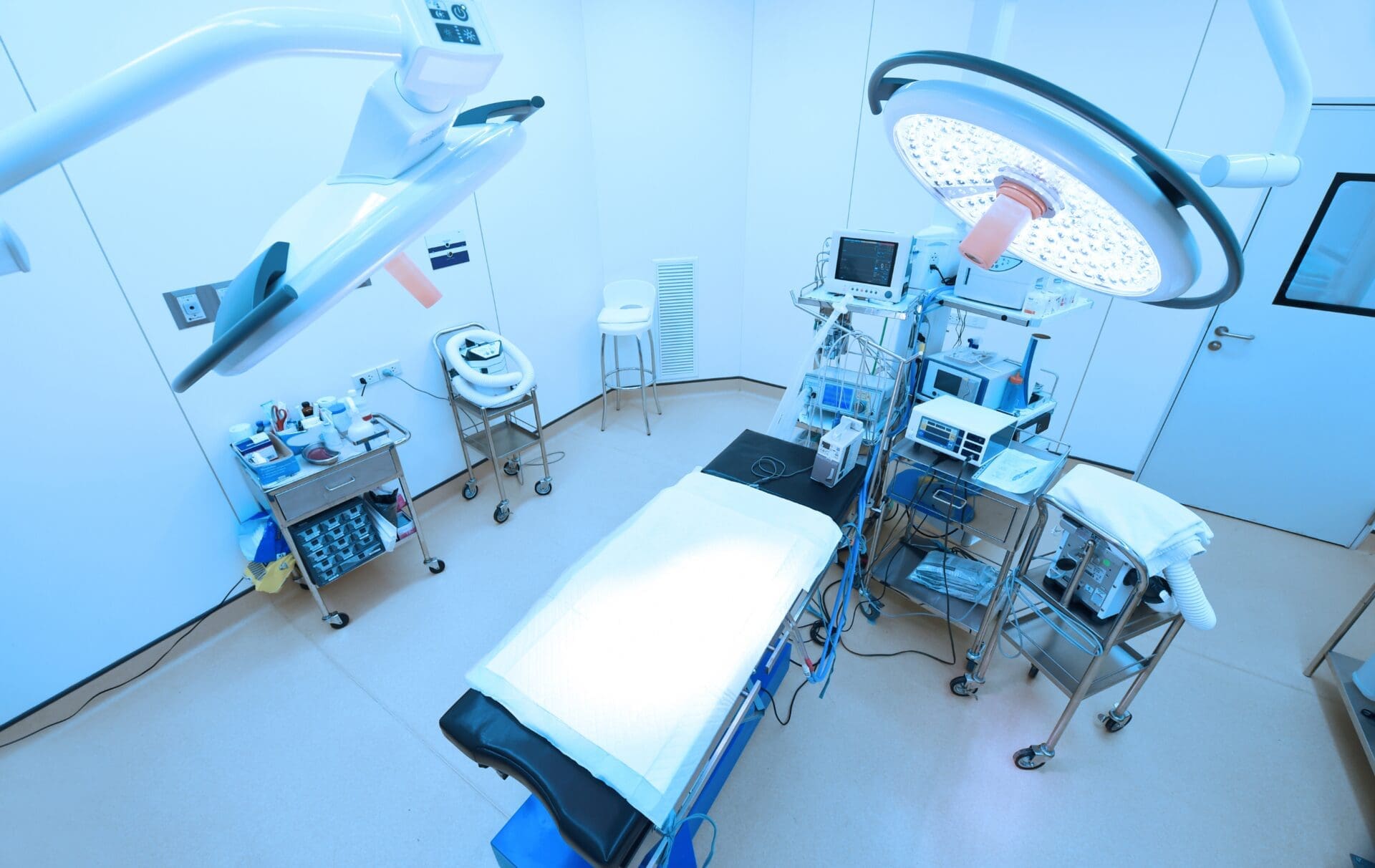
Complying with the rules on distance selling of medical devices and in vitro diagnostics device is a fundamental step when a manufacturer chooses to sell the devices online.

We are thrilled to announce that Obelis Group has been chosen as one of the regulatory consultants for the Luxemburgish Ministry of Economy’s Fit 4 Innovation

The US government issued the Modernisation of Cosmetics Regulation Act (MoCRA) of 2022, which will apply as of December 29, 2023.
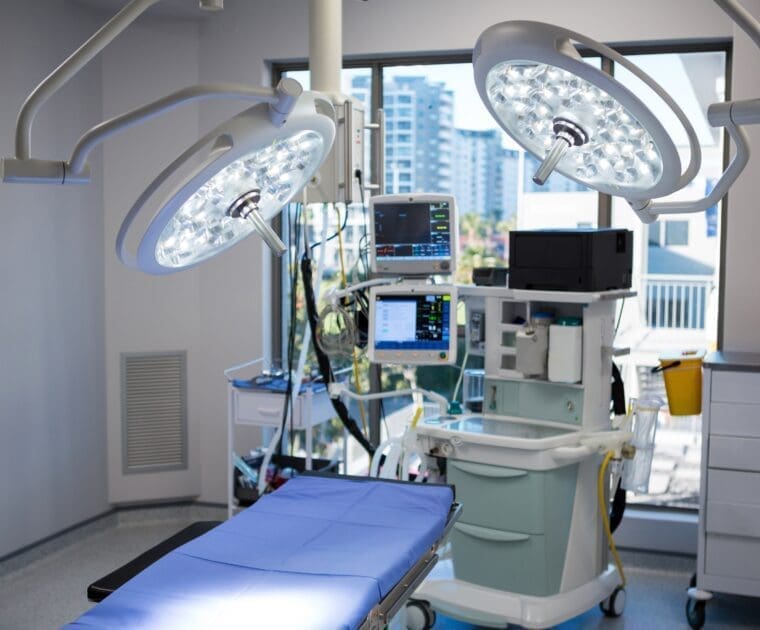
Medical Device and In-vitro Diagnostic Industry Discussion: Key Takeaways on IVDR and MDR Implementation Challenges

To prevent market disruptions and give manufacturers enough time to certify their medical devices under…

The new EU Regulation on in vitro diagnostic devices (IVDR), applicable since May 26, 2022, affected the classification and compliance process of COVID-19 tests.