Welcome to the world of cosmetics, where labelling compliance is the key to unlocking market…


Welcome to the world of cosmetics, where labelling compliance is the key to unlocking market…

In the ever-evolving cosmetics industry, achieving compliance with regulations is paramount. However, juggling multiple regulations…
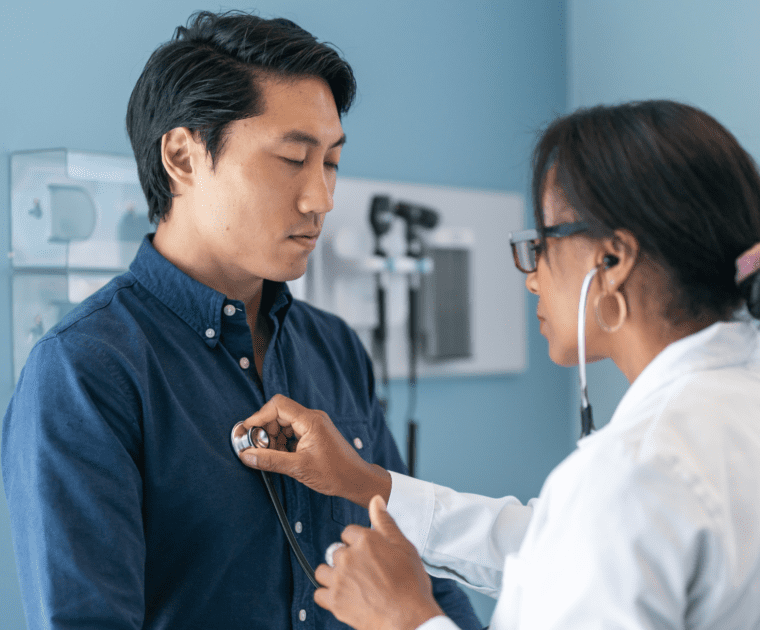
As an IVD (In Vitro Diagnostic) Korean manufacturer, accessing the EU (European Union) market can…

Most in vitro diagnostic medical devices require the involvement of a notified body or approved…

Register to our new webinar to learn about the MDR Legacy extension in EU, UK and CH. Register now!

Introducing new restrictions for Butylated Hydroxytoluene (BHT) and Acid Yellow 3 in cosmetic products, the Commission Regulation (EU) 2022/2195 brings changes to the industry

Discover the new GDPR obligations for businesses and organizations within the EU and UK!

Discover the new GDPR obligations for businesses and organizations within the EU and UK!

On 23 May 2023 the Official Journal of the EU published a new law, the General Product Safety Regulation (GPSR). From 13 December 2024, manufacturers will have to comply with the new GPSR.

MoCRA deadlines you need to keep in mind when selling cosmetic products in the U.S