To modernise the European Standardisation System, the European Commission proposed to amend the Standardisation Regulation as part of its European Standardisation Strategy of February 2, 2022. Read all the details in our article!


To modernise the European Standardisation System, the European Commission proposed to amend the Standardisation Regulation as part of its European Standardisation Strategy of February 2, 2022. Read all the details in our article!

There are few direct implications to the Customes Agreements for manufacturer of medical devices from Turkey. Read all the details in our article!
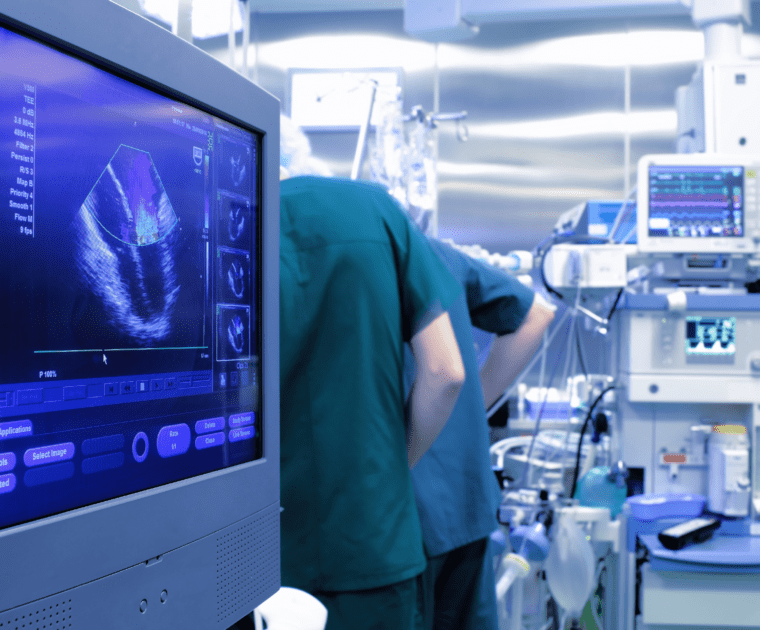
Vigilance and Post-Market Surveillance definition, differences and their scope. Visit our webpage for all the details!

The European Commission issued a proposal to restrict microplastics. The regulation will have an impact on products containing those substances. Read more!

Discover the effects of the implementation of MDR on the EU medical device market and the most relevant challenges in our new article!

Medical devices that have obtained CE Certificate under the MDD 93/42/EEC can circulate in the EU market. Learn the conditions here!

Join our latest Webinar about the EU IVDR vs. the Swiss IVDO.
Watch the recording to discover all the details from our expert!

On June 27, 2022, the Health Products Regulatory Authority (HPRA) published an alert about the serious health risks linked to the substance Melanotan 2. Read all the details!

The Spanish competent authority has updated its instructions for requesting authorisation for commercialising personal care products. Visit our page for more details!

If you are unsure of how you should now proceed in order to import new devices or maintain the previous ones in the European territory, this webinar is what you were looking for! Register now!